 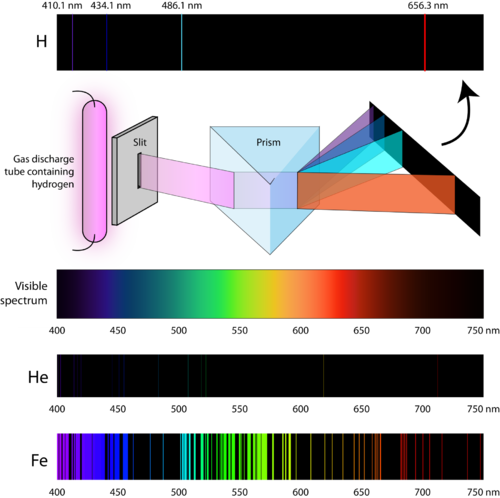
This type of spectrum is called an absorption spectrum.įor atoms of a particular element, there are usually more than one absorption line present because a ground state electron can be excited to different excited energy states by absorbing different amounts of energy (in the form of light). Therefore, absorption of light during electronic excitation causes there to be absent frequencies/wavelengths of light in the otherwise continuous spectrum. These absent lines are called absorption lines. When the same source of light is passed through atoms, ground state electrons can absorb specific amounts of energy to transition to higher energy states.ĭifferent energy of light corresponds to different frequencies/wavelengths. When white light is dispersed through a glass prism, it forms a continuous spectrum. White light consists of visible light of all wavelengths. Since atoms have multiple excitation states, there may be multiple electronic transitions, causing the emitted electromagnetic radiation to vary in energy and thus have different frequencies and wavelengths. When electrons return from their excited state to the ground state, energy is released in the form of electromagnetic radiation (EMR).ĭuring this process, electrons will release the same amount of energy as they absorbed (law of conservation of energy). 
If the energy provided does not exactly match this energy difference, it is does not absorbed by the electron.Įxcited electrons do not remain in their excited energy states permanently. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
